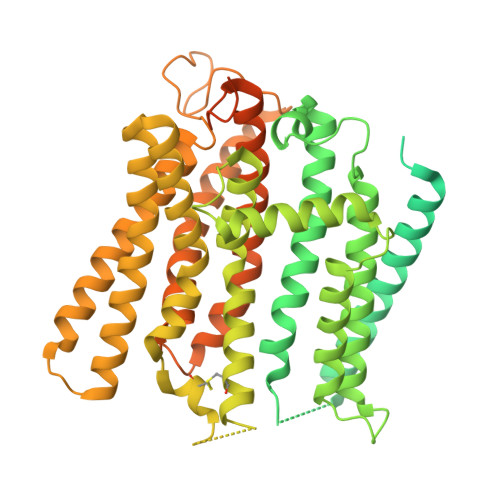
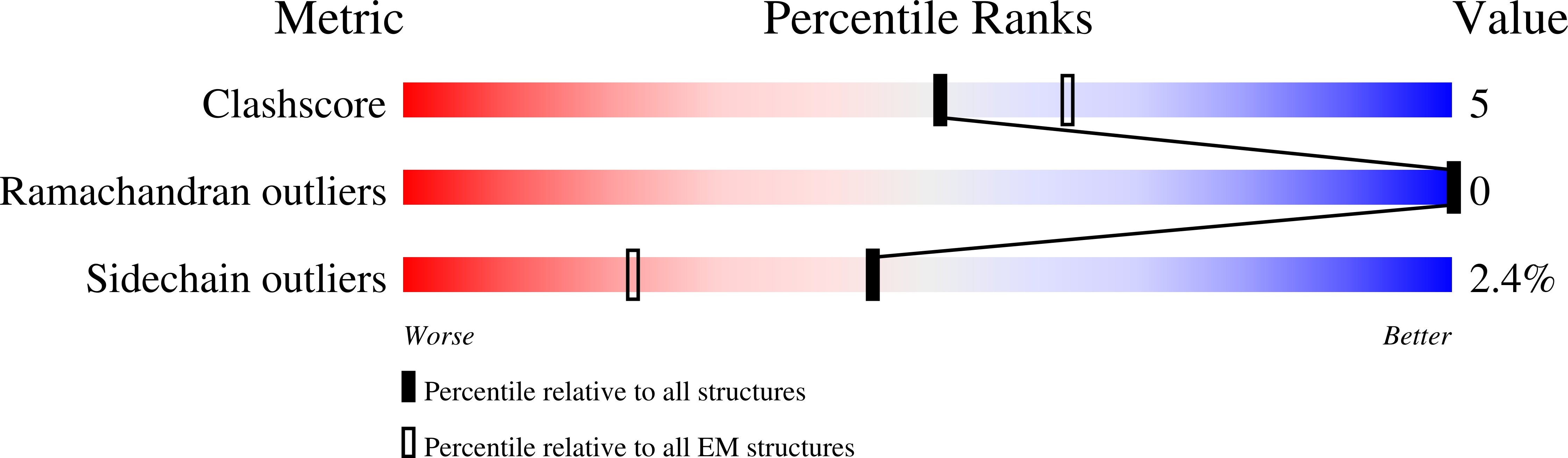Transport and InsP 8 gating mechanisms of the human inorganic phosphate exporter XPR1.
Zhu, Q., Yaggi, M.F., Jork, N., Jessen, H.J., Diver, M.M.(2025) Nat Commun 16: 2770-2770
- PubMed: 40113814
- DOI: https://doi.org/10.1038/s41467-025-58076-y
- Primary Citation of Related Structures:
9DVJ, 9DVK, 9DVL, 9DVM, 9DVN, 9DVO, 9DVP - PubMed Abstract:
Inorganic phosphate (Pi) has essential metabolic and structural roles in living organisms. The Pi exporter, XPR1/SLC53A1, is critical for cellular Pi homeostasis. When intercellular Pi is high, cells accumulate inositol pyrophosphate (1,5-InsP 8 ), a signaling molecule required for XPR1 function. Inactivating XPR1 mutations lead to brain calcifications, causing neurological symptoms including movement disorders, psychosis, and dementia. Here, cryo-electron microscopy structures of dimeric XPR1 and functional characterization delineate?the substrate translocation pathway and how InsP 8 initiates Pi transport. Binding of InsP 8 to XPR1, but not the related inositol polyphosphate InsP 6 , rigidifies the intracellular SPX domains, with InsP 8 bridging the dimers and SPX and transmembrane domains. Locked in this state, the C-terminal tail is sequestered, revealing the entrance to the transport pathway, thus explaining the obligate roles of the SPX domain and InsP 8 . Together, these findings advance our understanding of XPR1 transport activity and expand opportunities for rationalizing disease mechanisms and therapeutic intervention.
Organizational Affiliation:
Structural Biology Program, Memorial Sloan Kettering Cancer Center, New York, New York, USA.